|
The factor of a particular atom to the electronic construction of a compound is disguised by wave function-based quantum technicians.Scanlon, and C. Richard A.Catlow See Author Information Section of Components, Imperial College London, Birmingham SW7 2AZ, United Kingdom Départment of Chemistry, Univérsity College London, Lóndon WC1H 0AJ, United Kingdom Diamond Light Source Ltd., Gemstone House, Harwell Technology and Innovation Campus, Didcot, Oxfordshire OX11 0DAt the, United Empire Email: email protected Report this: L.Phys. Chem. Lett.
Publication Date (Web): Might 4, 2017 Distribution History. These metrics are usually regularly updated to reflect usage major up to the final few days. Citations are the amount of some other content citing this content, computed by Crossref and up to date daily. The Altmetric Interest Score is certainly a quantitative gauge of the attention that a analysis article has obtained online. Chemical Bonding In Solids Burdett Download A PagePressing on the donut image will download a page at altmetric.com with additional information about the score and the interpersonal media presence for the given article. Find more details on the Altmetric Interest Rating and how the rating is computed. IUPAC defines it as the charge an atom might end up being thought to possess when electrons are usually counted relating to an agreed-upon set of rules. Once the structure of a substance is recognized, a trained chemist will instantly infer the oxidation areas of its components, and in switch foresee the structural, digital, optical and permanent magnet qualities of the materials. In the contemporary era of quantum biochemistry, can the use of formal oxidation claims be justified Allow us consider the illustration of TiO 2, a broadly studied metallic oxide. Right after the standard guidelines, an oxidation condition of Ti(IV) is usually assigned. It may consequently be astonishing that a cost condition of Ti 2.5 in TiO 2 can be assigned on the time frame of current density functional concept (DFT) computations. The electron thickness in solids is routinely calculated using a variety of techniques in computational chemistry and tested in diffraction experiments. However, the cost on a provided ion is certainly not an observable, but relies on a choice of design or concept to partition the electron denseness between atomic facilities, and right now there will be in common no unambiguous method to do such dividing. The influx functions of electrons in crystals are multicentered and delocalized (Bloch waves) mainly because needed by quantum mechanics. Charges could become assigned unambiguously for the special situation that electron thickness does not overlap, which is, nevertheless, uncommon a point investigated in details within the evaluation by Catlow and Stoneham. Through topological analysis of the electron density, and by parting into atom-centered basins making use of the structure suggested by Bader (7) (one of several partition strategies (8) ), a partial charge of Ti 2.5 will be designated in TiO 2. One issue with Baders method is definitely that atoms or ions perform overlap. Partial costs such as these are peculiar quantities in hormone balance as oxidation and decrease processes involve modifications in integral figures of electrons. Polarization of án anion toward á cation is usually impossible to distinguish from a charge transfer on developing a heteropolar bond between like two ions. We notice, nevertheless, that relative modifications in partial charges (in similar chemical conditions) have got application for probing chemical processes, y.g., surface area catalytic responses. What can become measured In solid-state thermochemistry, charges matching to official oxidation expresses are constant with lattice energies from thermochemical data (BornHaber process) for a plethora of inorganic compounds. The response to electromagnetic fields is determined by the dielectric screening, which can end up being accurately defined by versions of polarizable ions with official charges, mainly because can the interatomic forces and hence the phonon distribution, (6) and such an assignment has equivalent validity with those structured on determined or sized charge densities. The unambiguous assignment of oxidation claims can become made from tests, e.gary the gadget guy., core-level photoemission spectroscopy, structured on guide to the matching ions in (usually aqueous) options. Furthermore, electrochemical tests allow one to count number charges going to electrodes straight, proving the (composite) actuality of formal oxidation areas, while optical strategies can motivate electronhole sets with particular degrees of localization. For any compound formulated with two or more components with differing electronegativity, the binding is heteropolar, which can be described using the language of covalency (i.elizabeth., hybridization of orbitaIs) or iónicity (i.elizabeth., polarization of ions). These are two alternative explanations of the exact same reality. From an ionic perspective, moving from a Ti0 2 molecule to the solid, we perform not modify the oxidation state, but rather improve the polarity of a genuine. As the coordination of air increases, the Madelung potential stabilizes the oxide anion, growing the ionization potential of the crystal clear. 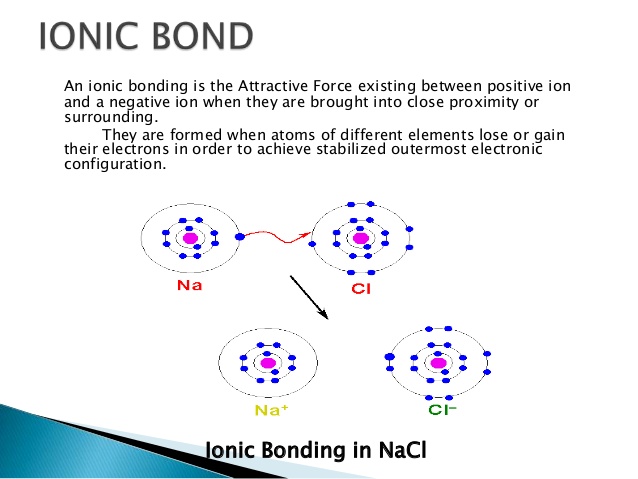
Electron keeping track of in solids is more challenging than it would very first appear.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
- Blog
- Safar ki dua in english and arabic
- Dearly beloved synthesia
- Sako riihimaki l46 -222
- Memorex cd label maker download free software
- Auxiliary and modal verbs exercises
- 1976 ome banjo
- Adobe photoshop cs2 keygen
- How to install ps2 emulator on android reddit
- Navcoder activation key
- Nba live 18 logo
- Hp deskjet f4180 driver software
- Mohabbatein full movie with eng subtitles
 RSS Feed
RSS Feed
